 Textbook content produced by OpenStax is licensed under a Creative Commons Attribution License. Determine the mass of iron and the volume of chlorine gas (measured at 25oC and 1 atm) that is. We recommend using aĪuthors: Paul Peter Urone, Roger Hinrichs Amps, time, Coulombs, Faradays, and moles of electrons. With Coulombs Law, we can use the +1 charge on Na+, the -1 charge on Cl. Use the information below to generate a citation. Coulombs Law Examples, and the Potential Energy Diagram for Hydrogen. Then you must include on every digital page view the following attribution: If you are redistributing all or part of this book in a digital format, Then you must include on every physical page the following attribution: If you are redistributing all or part of this book in a print format, For color-charged particles like quarks and hypothetical leptoquarks, the charge number is a multiple of 1/3.Want to cite, share, or modify this book? This book uses the In particle physics, the charge number is a (derived) flavor quantum number. So, electrons get shared between the two ions. 3) Al3+ is highly charged species in nature and it can polarize the electron clouds of Cl to a large extent. to make the ionic compound of NaCl: sodium chlorine, table salt. 2) One example of covalent bond is AlCl3. The unit of electric charge is the coulomb. In chemistry, the same charge numbers are usually indicated as superscript "+2" or "−2".Ĭharge numbers in elementary-particle physics 1) Aluminum is an only Amphoteric metal element that has the ability to form both ionic and covalent bond. some delta baryons) are indicated with a superscript "++" or "−−". Coulombs law works well for predicting the energy of an electron in a hydrogen atom (H has only two particles: one nucleus and one electron). Unlike in chemistry, subatomic particles with electric charges of two elementary charges (e.g. According to Coulombs law, the attraction of an electron to a nucleus depends only on three factors: the charge of the nucleus (+Z), the charge of the electron (-1), and the distance between the two (r). If the charge was positive, an electron would be lost and taken away.Ĭharge numbers in nuclear and hadron physics įor an atomic nucleus, which can be regarded as an ion having stripped off all electrons, the charge number is identical with the atomic number Z, which corresponds to the number of protons in ordinary atomic nuclei. Since there is a negative charge on the outside of the Lewis dot structure, one electron needs to be added to the structure. For example, if the structure is an ion, the charge will be included outside of the Lewis dot structure. What is the charge in coulombs of the nucleus of a chlorine atom Question: What is the charge in coulombs of the nucleus of a chlorine atom Coulomb: Coulomb is an SI unit of electric. 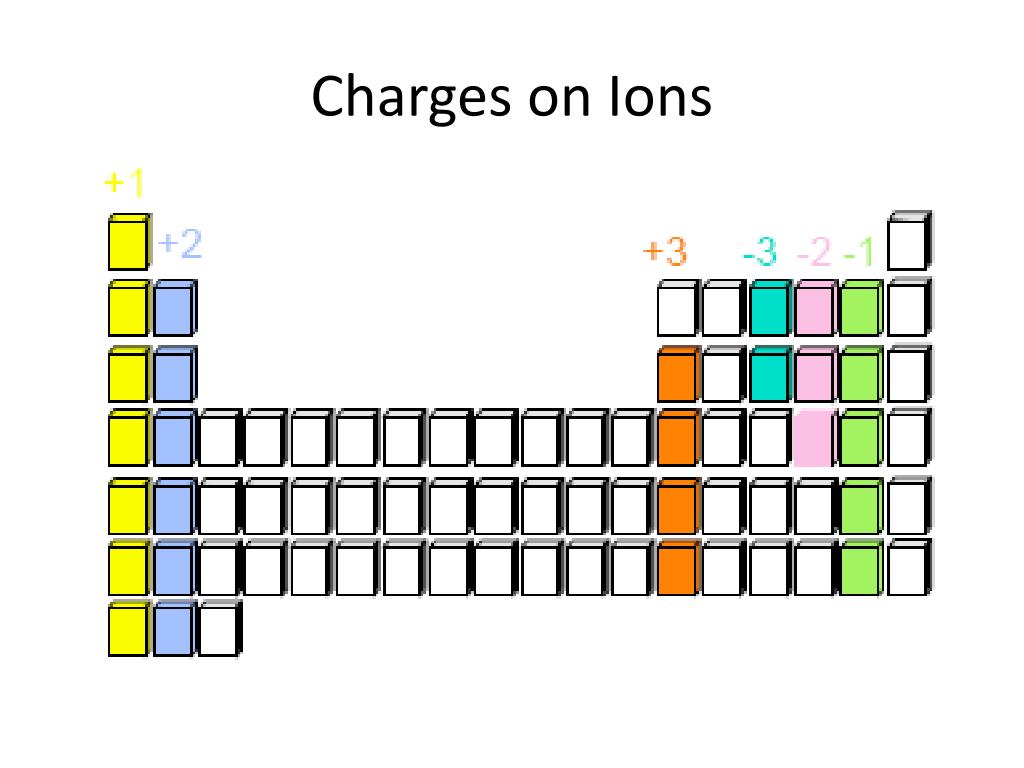
This helps when trying to solve oxidation questions.Ī charge number also can help when drawing Lewis dot structures. įor example, the charge on a chloride ion, C l − is +1. All particles of ordinary matter have integer-value charge numbers, with the exception of quarks, which cannot exist in isolation under ordinary circumstances (the strong force keeps them bound into hadrons of integer charge numbers).Ĭharge number or valence of an ion is the coefficient that, when multiplied by the elementary charge, gives the ion's charge. Atomic numbers ( Z) are a special case of charge numbers, referring to the charge number of an atomic nucleus, as opposed to the net charge of an atom or ion. 
Na + is a sodium ion with charge number positive one (an electric charge of one elementary charge). The charge numbers for ions (and also subatomic particles) are written in superscript, e.g. ( January 2014)Ĭharge number ( z) refers to a quantized value of electric charge, with the quantum of electric charge being the elementary charge, so that the charge number equals the electric charge ( q) in coulombs divided by the elementary-charge constant ( e), or z = q/ e. This article may need to be rewritten to comply with Wikipedia's quality standards. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
